 Abstract-The bark beetle, Pityogenes bidentatus (Coleoptera:Scolytidae), searches in mixed conifer and
deciduous forests of northern Europe for suitable branches
of its host, Scots pine (Pinus sylvestris).We tested whether odors from several diverse nonhost trees and plants
common in the habitat (e.g., mountain ash, Sorbus aucuparia; oak, Quercus robur; alder buckthorn, Frangula
alnus; blueberry, Vaccinium myrtillus; raspberry, Rubus idaeus; and grass, Deschampsia flexuosa)
would reduce the attraction of the bark beetle to traps releasing its aggregation pheromone components in the field.
Volatiles from the leaves or bark of each of these plants significantly reduced the attraction of the beetles
to their pheromone. Odors collected from these nonhosts and analyzed
by GC/MS contained monoterpenes, sesquiterpenes, and
"green-leaf "alcohols, several of which (e.g.,1-octene-3-ol and B-caryophyllene) reduced the attraction to pheromone
in the field and elicited electroantennographic responses. In the laboratory, reproduction by the beetle was
marginal in nonhost Norway spruce, Picea abies, and was
absent in the other nonhost trees. Olfactory avoidance of
unsuitable nonhosts may have evolved due to advantages
in avoiding mistakes during host selection.
Abstract-The bark beetle, Pityogenes bidentatus (Coleoptera:Scolytidae), searches in mixed conifer and
deciduous forests of northern Europe for suitable branches
of its host, Scots pine (Pinus sylvestris).We tested whether odors from several diverse nonhost trees and plants
common in the habitat (e.g., mountain ash, Sorbus aucuparia; oak, Quercus robur; alder buckthorn, Frangula
alnus; blueberry, Vaccinium myrtillus; raspberry, Rubus idaeus; and grass, Deschampsia flexuosa)
would reduce the attraction of the bark beetle to traps releasing its aggregation pheromone components in the field.
Volatiles from the leaves or bark of each of these plants significantly reduced the attraction of the beetles
to their pheromone. Odors collected from these nonhosts and analyzed
by GC/MS contained monoterpenes, sesquiterpenes, and
"green-leaf "alcohols, several of which (e.g.,1-octene-3-ol and B-caryophyllene) reduced the attraction to pheromone
in the field and elicited electroantennographic responses. In the laboratory, reproduction by the beetle was
marginal in nonhost Norway spruce, Picea abies, and was
absent in the other nonhost trees. Olfactory avoidance of
unsuitable nonhosts may have evolved due to advantages
in avoiding mistakes during host selection.
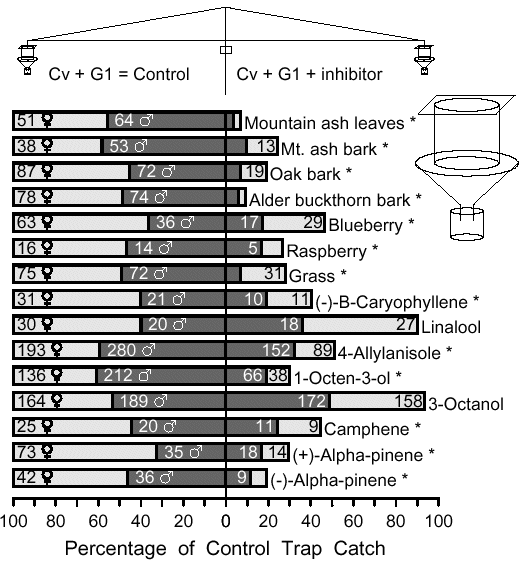 Fig.1 Catch of Pityogenes bidentatus on traps releasing pheromone
plus inhibitor volatiles compared to control traps with pheromone alone.
The trap pairs were mechanically-rotated at 2 rph to
minimize catch variation due to trap position.Test replicates were
conducted for at least 1 h, and after each replicate, the inhibitory
source, but not the attractants, was switched to the other trap.
Replicate catches were summed and the paired control and treat-
ment were compared with a X2 goodness of fit test to an expected
catch if there were no differences based on the average for both
traps. Tests used about 100 g (fresh weight)of bark strips or leaves/twigs
(9-12 May 2001), and the chemicals as indicated (27 April - 26 May 2001) near
Sjöbo, Sweden. P. bidentatus pheromone
components, cV [(S)-cis-verbenol, 96%, Borregaard) and G1
[racemic cis-configured grandisol, (1R*, 2S*)-1-(2-hydroxyethyl)-1-methyl-2-(1-methylethenyl)cyclobutane;
Frank Enterprises, Columbus, Ohio], were
released from small tubes at 21 and 0.5-2 ug/h,
respectively. (-)-trans-B-Caryophyllene (>99%, Sigma), linalool
(97%, Aldrich), 4-allylanisole (98%, Aldrich), 1-octen-3-ol (>99%,
ACROS), and 3-octanol (98%, ACROS) were released at 20, 160,
270, 230, and 200 ug/h, respectively. The "monoterpenes ", (-)-a-
pinene (>99.5%, Fluka), (+)-a-pinene (>99%,Fluka), and
camphene (99%, Aldrich) were released alone at about 1400,
1400, and 1075 ug/h, respectively, from various combinations of
tubes. Catches with asterisks were significantly lower than the
pheromone controls in the same test at P<0.01 (X2 goodness of fit)
Fig.1 Catch of Pityogenes bidentatus on traps releasing pheromone
plus inhibitor volatiles compared to control traps with pheromone alone.
The trap pairs were mechanically-rotated at 2 rph to
minimize catch variation due to trap position.Test replicates were
conducted for at least 1 h, and after each replicate, the inhibitory
source, but not the attractants, was switched to the other trap.
Replicate catches were summed and the paired control and treat-
ment were compared with a X2 goodness of fit test to an expected
catch if there were no differences based on the average for both
traps. Tests used about 100 g (fresh weight)of bark strips or leaves/twigs
(9-12 May 2001), and the chemicals as indicated (27 April - 26 May 2001) near
Sjöbo, Sweden. P. bidentatus pheromone
components, cV [(S)-cis-verbenol, 96%, Borregaard) and G1
[racemic cis-configured grandisol, (1R*, 2S*)-1-(2-hydroxyethyl)-1-methyl-2-(1-methylethenyl)cyclobutane;
Frank Enterprises, Columbus, Ohio], were
released from small tubes at 21 and 0.5-2 ug/h,
respectively. (-)-trans-B-Caryophyllene (>99%, Sigma), linalool
(97%, Aldrich), 4-allylanisole (98%, Aldrich), 1-octen-3-ol (>99%,
ACROS), and 3-octanol (98%, ACROS) were released at 20, 160,
270, 230, and 200 ug/h, respectively. The "monoterpenes ", (-)-a-
pinene (>99.5%, Fluka), (+)-a-pinene (>99%,Fluka), and
camphene (99%, Aldrich) were released alone at about 1400,
1400, and 1075 ug/h, respectively, from various combinations of
tubes. Catches with asterisks were significantly lower than the
pheromone controls in the same test at P<0.01 (X2 goodness of fit)
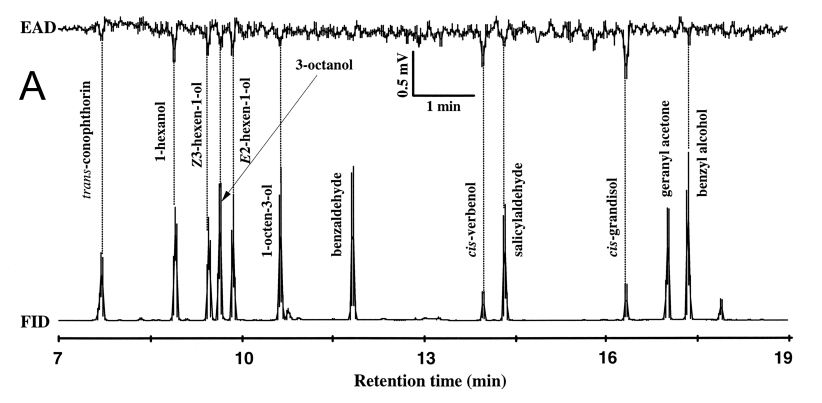
| Table 1 Release rates of volatiles (ug/h) from plant materials (leaves and bark of nonhosts mountain ash, Sorbus aucuparia; grass, Deschampsia flexuosa; raspberry, Rubus idaeus; and alder buckthorn, Frangula alnus; used in the field test of Pityogenes bidentatus inhibition to aggregation pheromone (9 - 12 May 2001, Fig.1). The volatiles were collected on Porapak Q and identified by GC/MSDa | |||||
| Compound | Mountain ash: S. aucuparia | Grass: D. flexuosa | Raspberry: R. idaeus | Alder Buckthorn: F. alnus | |
|---|---|---|---|---|---|
| - | Leaves | Bark | - | Leaves | Bark |
| a-Pinene | 0.15 | 0.1 | 0.3 | 0.05 | - |
| Camphene | -0.08 | - | - | - | - |
| B-Pinene | - | - | 0.06 | -- | - |
| 3-Carene | - | - | 0.09 | - | - |
| a-Ocimene | 0.06 | - | - | - | - |
| B-Ocimene | 1.4 | - | - | - | - |
| (E)-4,8-Dimethyl-1,3,7-nonatriene | 0.6 | - | - | - | - |
| Pentyl acetate | - | - | 0.07 | - | - |
| Z-2-Pentenyl acetate | - | - | 0.2 | - | - |
| Hexyl acetate | - | - | 1.3 | - | - |
| (Z)-3-Hexenyl acetate | 0.2 | - | 42 | 0.2 | - |
| (E)-2-Hexenyl acetate | - | - | 0.5 | - | - |
| 6-Methyl-5-hepten-2-one | - | - | 0.04 | - | - |
| (Z)-3-Hexenyl butanoate | 0.04 | - | - | - | - |
| 1-Hexanol | - | 0.06 | 0.1 | - | - |
| (Z)-3-Hexen-1-ol | - | - | 2.8 | - | - |
| 1-Octen-3-ol | - | - | 0.09 | - | - |
| Isopentyl proprionate | - | - | 0.1 | - | - |
| 3-Ethyl-3-pentanol | - | - | - | 0.05 | 0.07 |
| Nonanal | - | - | - | - | 0.05 |
| Benzaldehyde | 0.07 | 0.9 | - | - | - |
| Benzyl alcohol | - | 0.09 | - | - | - |
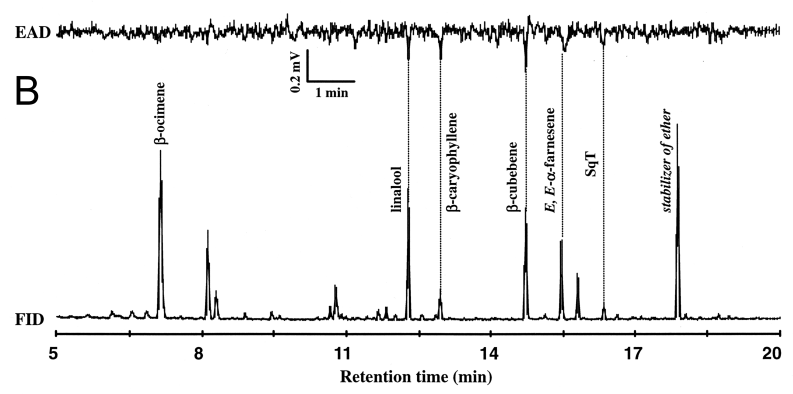
| Linalool | 0.7 | - | - | - | - |
| B-Bourbonen | 0.07 | - | - | - | - |
| B-Caryophelene | 0.2 | - | - | - | - |
| a-Cubebene | 0.1 | - | - | - | - |
| B-Cubebene | 1.0 | - | - | - | - |
| a-Farnesene | 0.5 | - | - | - | - |
| Methyl salicylate | 0.4 | - | - | - | - |
|
J.A. Byers1, Q.-H. Zhang Department of Crop Science Swedish University of Agricultural Sciences 230 53 Alnarp, Sweden G. Birgersson Chemical Ecology Göteborg University 405 30 Göteborg, Sweden Present addresses: 1J.A. Byers, Western Cotton Research Laboratory, USDA-ARS, 4135 E Broadway Road, Phoenix, AZ 85040, USA Q.-H. Zhang, Chemicals Affecting Insect Behavior Laboratory, USDA-ARS, BARC-West, B-007, 10300 Baltimore Avenue, Beltsville, MD 20705, USA G. Birgersson, Chemical Ecology and Toxicology, Lund University, 223 62 Lund, Sweden |
|---|