5.5.1 Precursors of aggregation pheromone components
Aggregation pheromone components were first identified in bark
beetles from male Ips paraconfusus as a synergistic blend of (S)-(-
)-ipsenol, (S)-(+)-ipsdienol, and (4S)-cis-verbenol (Silverstein et
al. 1966, 1967; Wood et al. 1968). Several other Ips species were
soon discovered to produce and respond to various blends of these
compounds (Vité et al., 1972). The similarity of chemical structure
between a major host monoterpene, myrcene, and ipsenol and
ipsdienol (Figs. 4 and 5) led Hughes (1974) to propose that in Ips
myrcene was a precursor of these pheromone components. Exposure of
males of I. paraconfusus to myrcene vapor resulted in their
production of compounds with GC retention times identical to
ipsenol and ipsdienol (Hughes, 1974). Byers et al. (1979) confirmed
these identifications using gas chromatography and mass
spectrometry (GC-MS) and behavioral assays, and reported the male-
specific quantitative relationships between precursor vapor
concentration and pheromone products. Hendry et al. (1980)
radiolabelled myrcene and established the direct conversion of
myrcene to the pheromone components (Fig. 8).
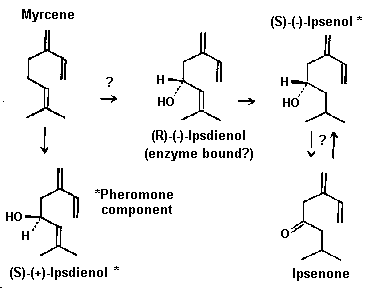 Fig. 8. Proposed scheme for the conversion of the host tree
compound, myrcene, to the pheromone components (S)-(-)-ipsenol and
(S)-(+)-ipsdienol in Ips paraconfusus based on radio-labelling
experiments and enantiomers found in the male (Silverstein et al.,
1966; Wood et al., 1968; Hughes, 1974; Renwick et al., 1976; Byers
et al., 1979; Fish et al., 1979; Hendry et al., 1980; Byers, 1981;
Byers and Wood, 1981). Conversion arrows with question marks have
not been proven. (R)-(-)-ipsdienol does not accumulate in the
hindgut but may occur as an enzyme-bound intermediate. However,
contrary to the scheme, the amounts of ipsenone, (S)-(-)-ipsenol
and (S)-(+)-ipsdienol in males were not correlated with myrcene
titers in the host trees.
Fig. 8. Proposed scheme for the conversion of the host tree
compound, myrcene, to the pheromone components (S)-(-)-ipsenol and
(S)-(+)-ipsdienol in Ips paraconfusus based on radio-labelling
experiments and enantiomers found in the male (Silverstein et al.,
1966; Wood et al., 1968; Hughes, 1974; Renwick et al., 1976; Byers
et al., 1979; Fish et al., 1979; Hendry et al., 1980; Byers, 1981;
Byers and Wood, 1981). Conversion arrows with question marks have
not been proven. (R)-(-)-ipsdienol does not accumulate in the
hindgut but may occur as an enzyme-bound intermediate. However,
contrary to the scheme, the amounts of ipsenone, (S)-(-)-ipsenol
and (S)-(+)-ipsdienol in males were not correlated with myrcene
titers in the host trees.
Earlier, Hughes
(1974) had proposed that ipsdienol was directly converted to
ipsenol, since topical application of ipsdienol on males resulted
in ipsenol production. This was confirmed by deuterium labelled
ipsdienol (64% d) being converted to labelled ipsenol (25% d; Fish
et al. 1979). Intermediates such as ipsdienone and ipsenone (Fig.
8) also have been suggested in the biosynthetic pathways (Fish et
al., 1979; Byers and Birgersson, 1990).
Another host monoterpene, (-)-alpha-pinene, in the vapor phase is
converted to (S)-cis-verbenol in both sexes of I. paraconfusus
(Fig. 9, Renwick et al. 1976), and the relationship between vapor
concentration and pheromone component production was quantified
(Byers 1981a).
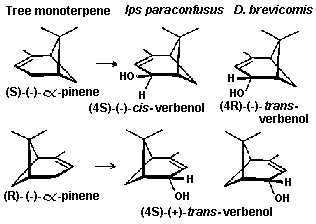 Fig. 9. Stereoselective conversion of the two enantiomers of the
host tree monoterpene, alpha-pinene, to pheromone components (4S)-cis-
verbenol in Ips paraconfusus and (4R)-trans-verbenol in D.
brevicomis.
Fig. 9. Stereoselective conversion of the two enantiomers of the
host tree monoterpene, alpha-pinene, to pheromone components (4S)-cis-
verbenol in Ips paraconfusus and (4R)-trans-verbenol in D.
brevicomis.
The production of (-)-cis-verbenol and (+)-trans-
verbenol in I. typographus and I. amitinus from the (-)- and (+)-
enantiomers of alpha-pinene was shown to have a similar relationship by
Klimetzek and Francke (1980). The ratio of cis-/trans-verbenols
produced by I. typographus was consistent with the ratio of
enantiomers of alpha-pinene in the host tree, which differed between
trees of different regions and genotypes (Klimetzek and Francke,
1980; Lindström et al., 1989).
Based on the above studies, a paradigm was established that I.
paraconfusus, and probably most other Ips species, use myrcene and
(-)-alpha-pinene in their host tree as precursors to ipsenol and
ipsdienol or cis-verbenol, respectively. The evolution of host
selection behavior by Ips bark beetles could be influenced by the
amounts of alpha-pinene and myrcene in the tree (Elkinton et al. 1980).
The variation of monoterpenes among trees within a species (Mirov
1961; Smith 1964, 1967) might allow beetles to have evolved
preferences for trees that had large amounts of precursor for use
in biosynthesis of aggregation pheromone components. A second
hypothesis is that tree genotypes may have evolved through natural
selection which are lower in pheromone precursor monoterpenes as a
means of resistance to bark beetles (Byers, 1989a). It is still not
known whether selection pressure by bark beetle predation is severe
enough to cause the evolution of trees with lower amounts of alpha-
pinene as a means of resistance.
There appears not to be any coevolution between myrcene
precursor from the host and its conversion by Ips paraconfusus to
certain pheromone components. This is because males of I.
paraconfusus produced almost identical amounts of the pheromone
components ipsenol and ipsdienol when feeding in five different
host pine species, regardless of the large differences in
concentrations of myrcene in the host-tree species (Byers and
Birgersson, 1990). In fact, digger pine, P. sabiniana, had so
little myrcene that it could not be detected by GC-MS, and it was
calculated that a beetle would need to eat at least eight times its
weight in toxic and repellent oleoresin in order to have any
possibility of obtaining the required amounts of myrcene.
Apparently in this case the expected energetic advantages of using
a precursor from the host that is structurally similar to the
pheromone components are outweighed by the need for the beetle to
retain control over the ecologically critical pheromone system. The
beetle could maintain control by means of either de novo
biosynthesis or use of a simpler host precursor found in all
potential hosts.
Streptomycin, a 70S ribosome-active antibiotic, when fed to I.
paraconfusus inhibited their ability to produce ipsenol and
ipsdienol, but the antibiotic had no affect on alpha-pinene conversion
to cis-verbenol (Byers and Wood, 1981b; Hunt and Borden, 1989).
However, I. paraconfusus reared axenically (without microorganisms)
could still produce reduced amounts of ipsenol and ipsdienol (Conn
et al., 1984; Hunt and Borden, 1989). Any symbiotic microorganisms
involved in pheromone biosynthesis would have to be transovarially
transmitted in order to survive the axenic rearing methods (Hunt
and Borden, 1989). Juvenile hormone is also implicated in pheromone
biosynthesis: topical application induces ipsenol and ipsdienol
production in I. paraconfusus in the absence of external myrcene
(Hughes and Renwick, 1977). Other pheromones such as verbenone,
exo-brevicomin and frontalin are found in emerged D. brevicomis
before landing on the tree D. brevicomis (Byers et al.,
1984).
Exposure of Ips duplicatus to myrcene vapor causes the beetle
to produce small amounts of its pheromone component ipsdienol but
not E-myrcenol, the other component (Schlyter et al., 1992). Both
compounds are expected to be made from myrcene (Pierce et al.,
1987). Ivarsson et al. (1993) found that the biosynthesis of both
E-myrcenol and ipsdienol from mevalonate in I. duplicatus could be
blocked by the enzyme inhibitor compactin, but the production of
cis-verbenol from alpha-pinene in I. typographus was not affected by
the inhibitor. These studies, and those on I. paraconfusus,
indicate that the major pathway in nature for biosynthesis of the
pheromone components ipsdienol, ipsenol, and E-myrcenol in Ips is
de novo from mevalonate.

Byers, J.A. 1995. Host tree chemistry affecting colonization in bark
beetles, in R.T. Cardé and W.J. Bell (eds.). Chemical Ecology of
Insects 2. Chapman and Hall, New York, pp. 154-213.
 Fig. 8. Proposed scheme for the conversion of the host tree
compound, myrcene, to the pheromone components (S)-(-)-ipsenol and
(S)-(+)-ipsdienol in Ips paraconfusus based on radio-labelling
experiments and enantiomers found in the male (Silverstein et al.,
1966; Wood et al., 1968; Hughes, 1974; Renwick et al., 1976; Byers
et al., 1979; Fish et al., 1979; Hendry et al., 1980; Byers, 1981;
Byers and Wood, 1981). Conversion arrows with question marks have
not been proven. (R)-(-)-ipsdienol does not accumulate in the
hindgut but may occur as an enzyme-bound intermediate. However,
contrary to the scheme, the amounts of ipsenone, (S)-(-)-ipsenol
and (S)-(+)-ipsdienol in males were not correlated with myrcene
titers in the host trees.
Fig. 8. Proposed scheme for the conversion of the host tree
compound, myrcene, to the pheromone components (S)-(-)-ipsenol and
(S)-(+)-ipsdienol in Ips paraconfusus based on radio-labelling
experiments and enantiomers found in the male (Silverstein et al.,
1966; Wood et al., 1968; Hughes, 1974; Renwick et al., 1976; Byers
et al., 1979; Fish et al., 1979; Hendry et al., 1980; Byers, 1981;
Byers and Wood, 1981). Conversion arrows with question marks have
not been proven. (R)-(-)-ipsdienol does not accumulate in the
hindgut but may occur as an enzyme-bound intermediate. However,
contrary to the scheme, the amounts of ipsenone, (S)-(-)-ipsenol
and (S)-(+)-ipsdienol in males were not correlated with myrcene
titers in the host trees. Fig. 9. Stereoselective conversion of the two enantiomers of the
host tree monoterpene, alpha-pinene, to pheromone components (4S)-cis-
verbenol in Ips paraconfusus and (4R)-trans-verbenol in D.
brevicomis.
Fig. 9. Stereoselective conversion of the two enantiomers of the
host tree monoterpene, alpha-pinene, to pheromone components (4S)-cis-
verbenol in Ips paraconfusus and (4R)-trans-verbenol in D.
brevicomis.