5.4 PLANT COMPOUNDS AND RESIN IN RESISTANCE MECHANISMS
Resistance of pines to bark beetle attacks has long been attributed
to the amount of resin exudation and formation of pitch tubes
(Webb, 1906). It is obvious that oleoresin acts to entrap and
impede the excavation efforts of bark beetles. Dead beetles can
often be seen in crystallized resin of pitch tubes.
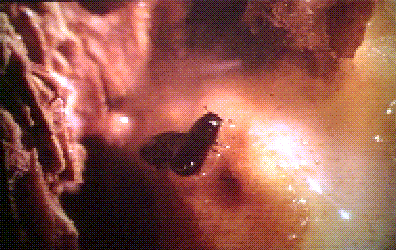 Fig. 7. Western pine beetles, Dendroctonus brevicomis, "swimming"
in oleoresin exuding from a "pitch tube" on the bark of ponderosa
pine. The beetle (in center of photo) and its mate underneath were
observed to burrow down through the viscous liquid into the
entrance of the gallery (directly under and to right of beetles)
for up to several minutes before returning and shoving out more
resin. The resin is slightly toxic and may exhaust the beetles; it
also may eventually crystallize to entrap them.
Fig. 7. Western pine beetles, Dendroctonus brevicomis, "swimming"
in oleoresin exuding from a "pitch tube" on the bark of ponderosa
pine. The beetle (in center of photo) and its mate underneath were
observed to burrow down through the viscous liquid into the
entrance of the gallery (directly under and to right of beetles)
for up to several minutes before returning and shoving out more
resin. The resin is slightly toxic and may exhaust the beetles; it
also may eventually crystallize to entrap them.
However, D.
brevicomis and other aggressive bark beetles have a great ability
to survive the "toxic" monoterpenes and suffocating mucilage and
may struggle for hours in copious resin flows (D. frontalis, Hodges
et al., 1979). I have observed D. brevicomis beetles completely
covered with resin while attempting to clear the entrance tunnel
(Fig. 7), and it appears that "breaths" of air are taken by
slightly lifting the elytra that protect the spiracle openings on
the dorsal side of the abdomen; later these beetles entered the
tree and made galleries (Byers et al., 1984). In experiments where
three female D. ponderosae were caged on each of 79 trees, only 43
trees were attacked, and of these just 15 were colonized by
aggregating beetles (Raffa and Berryman, 1979).
Oleoresin may provide resistance to trees due to chemical
toxicity to the beetle and associated microorganisms or to physical
impedance and entrapment (Hodges et al., 1985). Oleoresin and the
monoterpenes therein are repellent to bark beetles in concentrated
amounts (Struble, 1957; Pitman et al., 1966; Berryman and Ashraf,
1970; Bordasch and Berryman, 1977). Drought and poor water balance
lowers the resistance of conifers (Hodges and Lorio, 1975; Hodges
et al., 1979) probably by lowering the turgidity of resin duct
cells which lowers the oleoresin exudation pressure (OEP). A
correlation between higher OEP and greater resistance of ponderosa
pine to attack by D. brevicomis and I. paraconfusus has been
reported (Vité, 1961; Wood and Vité, 1961; Wood, 1962; Brown et
al., 1987). Hodges et al. (1979) found that in more resistant pines
their resin was slower to crystalize (P. elliotii) or had a higher
resin flow (P. palustris) compared to more susceptible trees (P.
taeda and P. echinata) colonized by D. frontalis. Cook and Hain
(1987) also found that susceptible shortleaf pines, P. echinata,
had a lower resin flow than resistant trees. However, Raffa and
Berryman (1982b) found no relationship between resistance and the
rate of resin flow or crystallization. Also, Schroeder (1990) found
no difference in resin flow between resistant and susceptible Scots
pine, P. sylvestris, feed on by T. piniperda. Western larch, Larix
occidentalis, had no OEP but the trees with higher content of 3-
carene in the resin were attacked less by D. pseudotsugae (Reed et
al., 1986).
Another factor that might be more important for resistance
could be the toxicity of compounds within the resin. Smith (1961,
1965a, b) exposed beetles of several Dendroctonus species to resin
vapors from host and nonhost pines and found beetles were able to
tolerate vapors of their host better than nonhosts. In an attempt
to determine which components of the resin vapor were toxic to bark
beetles, various conifer monoterpenes were presented neat at vapor
saturation to D. brevicomis held in a glass chamber (Smith, 1965a).
The most toxic monoterpene was limonene, followed by (+)-3-carene,
myrcene, (-)-B-pinene and then alpha-pinene. In another study, Smith
(1965b) found that n-heptane, a major constituent of Jeffrey pine
(P. jeffreyi), was quite toxic to D. ponderosae which does not feed
in this tree, whereas Dendroctonus jeffreyi beetles showed little
mortality in saturated vapors. However, the monoterpenes when
presented alone at saturation were from 40 to 80 times higher in
concentration than in the natural atmosphere of a beetle's gallery,
as found empirically by GC headspace analysis and explained by
Raoult's law that states the vapor pressure of a compound is due to
its mole percentage in the substrate/solvent mixture (Byers,
1981a). Most of these species may also avoid monoterpene vapors
temporarily by breathing at "ventilation" holes through the outer
bark. Thus, Smith's conclusion that monoterpenes are toxic under
natural conditions is dubious, but his results may still indicate
these monoterpenes increase mortality of bark beetles over longer
periods during feeding and colonization. Raffa et al. (1985)
exposed Scolytus ventralis to the above monoterpene vapors and also
found limonene the most toxic, but myrcene, alpha-pinene, B-pinene, and
3-carene, in that order also caused significant mortality.
Differences in monoterpene composition within a tree are
slight, even from year to year (Smith, 1964; Byers and Birgersson,
1990). Differences in monoterpene ratios can be large between trees
of one area, almost as large as differences over wide geographic
regions (Smith, 1964, 1966, 1967, 1969). The oleoresins of 88 trees
under attack by D. brevicomis and subsequently killed were compared
to those from 202 living trees, and the living trees had a higher
content of myrcene and limonene in their resins (Smith, 1966). This
correlation supports the theory that limonene is important in host
resistance.
Sturgeon (1979) theorized that D. brevicomis is a selective
force in the evolution of P. ponderosa and that therefore areas of
recent outbreaks might have selected trees with a higher titer of
limonene (since these trees would be more resistant). Eight
populations of trees, a total of 617 trees, were sampled for
monoterpenes and analyzed by principal component multivariate
statistic. The average proportions of myrcene and alpha-pinene in
resins from the eight populations ranged the least (10.0-15.2% and
4.5-9.2%, respectively), while the other monoterpenes varied more
(3-carene, 23.6-60.4%; B-pinene, 13-35%; limonene, 6.1-18.7%). The
populations were separated by the Cascade Range in northern
California and southern Oregon into two regions. The west side had
higher proportions of myrcene, B-pinene, and limonene. The
proportion of limonene in resin from three populations considered
to have been historically under heavy bark beetle predation was
higher than in populations not considered to have such a history
(Sturgeon, 1979). The problem with these findings is that a
correlation is made between rather accurate monoterpene results and
rather poor knowledge of former predation pressure by D.
brevicomis, or for that matter other bark beetle species in each of
the areas.
Sturgeon (1979) concluded that D. brevicomis beetles (and
others) may exert a frequency-dependent selection pressure on
chemically polymorphic populations of ponderosa pine. Thus after
beetles have colonized most of the low limonene and less resistant
trees during an epidemic, the beetle population would either (1)
die out or disperse to areas that had more chemically susceptible
trees or (2) evolve a tolerance to limonene. The second possibility
was considered less likely because of the large variation in
monoterpene composition among trees that would make it improbable
that selection of beetles would occur that were capable of
detoxifying all of these compounds. However, bark beetles,
including I. paraconfusus and D. brevicomis, already must be able
to detoxify all of these compounds since they survive exposure to
monoterpene vapors in part by converting them to oxygenated
products that are more soluble and readily excreted (Hughes 1973,
1974; Byers, 1981a, b, 1982, 1983b, c; Pierce et al., 1987).
Another hypothesis is that limonene might not always be the most
toxic to a bark beetle population, but rather those monoterpenes
that the population is not well adapted to (since they occur
infrequently) are the most toxic. In this regard, lodgepole pines
in some regions of California have very high titers of limonene
(Byers and Birgersson, 1990), yet they are readily attacked and
killed by D. ponderosae.
Gollob (1980) measured the monoterpene content of resin from
unattacked loblolly pines, P. taeda. Two apparently resistant pines
that had survived attack by D. frontalis in an epidemic area had a
much higher content of myrcene compared to other trees that were
killed by the beetle and had low or trace amounts of myrcene.
However, no consistent differences in monoterpene composition of
Douglas-fir, P. menziesii var. glauca, resin were found between
trees that had resisted attack by D. pseudotsugae and trees that
had succumbed (Hanover and Furniss, 1966). Similarly, Raffa and
Berryman (1982b) found no relationship between monoterpene
composition and degree of resistance of lodgepole pines, P.
contorta var. Latifolia, to D. ponderosae. Hodges et al. (1979)
also did not find differences in monoterpene or resin acid
composition which could account for differences in resistance among
four pine species to attack by D. frontalis.
In addition to the wound or primary resin production, conifers
have a secondary or hypersensitive response to attack (Reid et al.,
1967; Berryman 1969, 1972; Berryman and Ashraf, 1970; Raffa and
Berryman, 1982c, 1983; Christiansen et al., 1987). The tree
responds by isolating the invading insect or fungus within a lesion
of dead cells and secondary resin by autolysis of cells and
formation of traumatic resin containing higher concentrations of
monoterpenes and phenolics (Reid et al., 1967, Berryman, 1969,
1972, Shrimpton, 1973, Wong and Berryman, 1977; Wright et al.,
1979; Raffa and Berryman, 1982b, c, 1983; Hain et al., 1983,
Croteau et al., 1987).
Croteau et al. (1987) identified elevated levels of
monoterpenes and diterpene resin acids in stems of lodgepole pine
inoculated with blue-stain fungus Ceratocystis clavigera, resulting
in induced lesions and secondary resin production. The inoculated
stems contained about three times more of the monoterpenes alpha-
pinene, B-pinene, 3-carene and B-phellandrene, but less limonene.
De novo resin synthesis was indicated in the infected tissue since
radiolabelled sucrose was incorporated up to 20 times faster into
monoterpenes and up to 10 times faster into diterpene resin acids.
Chitosan, a fungal wall fragment, induced monoterpene biosynthesis
and increased levels of terpene cyclase enzyme which converted
radiolabelled terpene precursors (geranyl pyrophosphate and
farnesyl pyrophosphate) to labelled monoterpenes and sesquiterpene
olefins, respectively (Croteau et al., 1987). Nearly all S.
ventralis females leave their entrance holes in grand fir, Abies
grandis, when hypersensitive reactions are evident (Berryman and
Ashraf, 1970). Shorter egg galleries are made or fewer eggs are
laid by females in lesions (Berryman and Ashraf, 1970; Paine and
Stephen, 1988).
The hypersensitive wound reaction not only affects bark
beetles directly, but inhibits symbiotic microorganisms from
growing and killing the tree. Blue-stain and other symbiotic fungi
are surrounded and isolated in the lesions (Wong and Berryman,
1977; Stephen and Paine, 1985; Paine and Stephen, 1987, 1988).
Sometimes, phytopathogenic fungi (e.g. Ceratocystis minor var.
barrasii) that are carried in the beetle's mycangium (D. frontalis)
do not stimulate the hypersensitive response and thus spread to
kill the tree (Paine and Stephen, 1987). Possibly the fungi secrete
compounds, such as water soluble glycans, that inhibit the plant's
defensive hypersensitivity - as in potatoes (Doke and Tomiyama,
1980). Cobb et al. (1968) cultured four species of Ceratocystis
fungi associated with bark beetles (species: ips, minor,
schrenkiana, and pilifera) as well as the root pathogen fungus,
Fomes annosus, during exposure to saturated atmospheres of
oleoresin or monoterpenes of the host ponderosa pine. They found
that all fungal species were inhibited in growth by oleoresin
except C. ips, while all the monoterpenes (the five discussed above
plus B-phellandrene and camphene) as well as undecane (present in
Jeffrey pines) inhibited growth of the fungal species. Camphene and
undecane were the least toxic, alpha-pinene was intermediate, while B-
pinene, B-phellandrene, and 3-carene were more toxic, with myrcene
and limonene the most toxic. However, the most toxic of all
compounds tested was n-heptane (also the most volatile), a major
constituent of Digger pine, P. sabiniana (Mirov, 1961). When the
monoterpenes were incorporated into the culture medium as well as
in the vapor phase, myrcene and B-phellandrene appeared the most
toxic to most species, but all of the monoterpenes and undecane
reduced fungal growth (Cobb et al., 1968). They also indicated that
(+)-alpha-pinene was more inhibitory than the (S)-enantiomer. The
growth inhibition by monoterpenes was proposed to allow the tree
time to synthesize phenols (Shain, 1967) such as pinosylvin
(Anderson, 1962) that would kill the fungi (Cobb et al.,
1968).
Bridges (1987) tested alpha-pinene and B-pinene on two mycangial
fungi of D. frontalis and the blue stain fungi, C. minor. B-pinene
inhibited growth of C. minor, both monoterpenes inhibited one
mycangial fungi while the other fungus was stimulated. A
phenylpropanoid, 4-allylanisole, from P. taeda resin inhibited
growth of all fungi tested. Himejima et al. (1992) steam distilled
ponderosa pine oleoresin into a distillate of monoterpenes and
sesquiterpenes and a residue of four diterpene acids. The
individual monoterpenes of the distillate were not inhibitory to
growth of several gram-positive bacteria (at 800 ćg/ml) but did
inhibit two species of fungi. Longifolene, a sesquiterpene,
inhibited the gram-positive but not the gram-negative bacteria.
Other species of common mold fungi were not affected by the
monoterpenes, but the diterpene acid, abietic acid, was effective
against three species of gram-positive bacteria. Although these
species of microorganisms are not those associated with bark
beetles, the results indicate that oleoresin constituents may be
important as a general defense against microorganisms.
The carbon balance of a tree is the relative level of
photosynthate available for growth, maintenance, and biosynthesis
of defensive compounds. Christiansen et al. (1987) have reasoned
that too little moisture (drought), insect defoliation, and root
pathogens will reduce the amounts of carbon available for
biosynthesis of primary and secondary resin. Mild drought may
actually increase resistance of trees by lowering growth rates and
shifting the use of photosynthate to the biosynthesis of defensive
chemicals, while extended drought will increase the probability of
bark beetle outbreaks due to the depleted carbon reserves
(Christiansen et al., 1987). A similar theory of tree resistance
and attack by southern pine beetles was developed by Lorio
(1986).

Byers, J.A. 1995. Host tree chemistry affecting colonization in bark
beetles, in R.T. Cardé and W.J. Bell (eds.). Chemical Ecology of
Insects 2. Chapman and Hall, New York, pp. 154-213.
 Fig. 7. Western pine beetles, Dendroctonus brevicomis, "swimming"
in oleoresin exuding from a "pitch tube" on the bark of ponderosa
pine. The beetle (in center of photo) and its mate underneath were
observed to burrow down through the viscous liquid into the
entrance of the gallery (directly under and to right of beetles)
for up to several minutes before returning and shoving out more
resin. The resin is slightly toxic and may exhaust the beetles; it
also may eventually crystallize to entrap them.
Fig. 7. Western pine beetles, Dendroctonus brevicomis, "swimming"
in oleoresin exuding from a "pitch tube" on the bark of ponderosa
pine. The beetle (in center of photo) and its mate underneath were
observed to burrow down through the viscous liquid into the
entrance of the gallery (directly under and to right of beetles)
for up to several minutes before returning and shoving out more
resin. The resin is slightly toxic and may exhaust the beetles; it
also may eventually crystallize to entrap them.