Byers, J.A. 2005. A cost of alarm pheromone production in cotton aphids, Aphis gossypii. Naturwissenschaften 92:69-72. 
 Abstract.
The sesquiterpene, (E)-B-farnesene, is used by many aphid species as an alarm pheromone to warn related individuals of predation. Disturbed cotton aphids, Aphis gossypii Glover, released (E)-B-farnesene into the air as detected by SPME (solid phase microextraction) and gas chromatography mass spectrometry (GC-MS). Solvent extracts of cotton aphids of various life stages and weights also were analyzed by GC-MS for sums of ions 69 and 93, which discriminated (E)-B-farnesene from coeluting compounds. Aphids of all life stages and sizes reared on cotton plants in both an environmental chamber and glasshouse contained (E)-B-farnesene in amounts ranging from 0.1 to 1.5 ng per individual. The quantities of (E)-B-farnesene in aphids increased in relation to increasing body weight, and variation in individual weights explained about 82% of the variation in alarm pheromone. However, the concentrations (ng/mg fresh weight) declined exponentially with increasing body weight. These findings indicate that aphid nymphs try to compensate by producing relatively more pheromone per weight than adults but still cannot approach an evolutionary optimal load, as assumed in adults with the greatest total amounts. This suggests that young aphids need to balance costs of growth and maturation with costs of producing the alarm pheromone.
Abstract.
The sesquiterpene, (E)-B-farnesene, is used by many aphid species as an alarm pheromone to warn related individuals of predation. Disturbed cotton aphids, Aphis gossypii Glover, released (E)-B-farnesene into the air as detected by SPME (solid phase microextraction) and gas chromatography mass spectrometry (GC-MS). Solvent extracts of cotton aphids of various life stages and weights also were analyzed by GC-MS for sums of ions 69 and 93, which discriminated (E)-B-farnesene from coeluting compounds. Aphids of all life stages and sizes reared on cotton plants in both an environmental chamber and glasshouse contained (E)-B-farnesene in amounts ranging from 0.1 to 1.5 ng per individual. The quantities of (E)-B-farnesene in aphids increased in relation to increasing body weight, and variation in individual weights explained about 82% of the variation in alarm pheromone. However, the concentrations (ng/mg fresh weight) declined exponentially with increasing body weight. These findings indicate that aphid nymphs try to compensate by producing relatively more pheromone per weight than adults but still cannot approach an evolutionary optimal load, as assumed in adults with the greatest total amounts. This suggests that young aphids need to balance costs of growth and maturation with costs of producing the alarm pheromone.
Introduction
The sesquiterpene, (E)-B-farnesene, is the alarm pheromone of aphid species in over 30 genera, including the cotton aphid Aphis gossypii Glover, (Kislow and Edwards 1972; Bowers et al. 1972; Edwards et al. 1973; Wientjens et al. 1973; Pickett and Griffiths 1980; Mondor and Roitberg 2000; El-Sayed 2004). An aphid grabbed by forceps, mimicking a predator, or by an actual predator will release droplets from its cornicles that contain (E)-B-farnesene (Kislow and Edwards 1972; Nault et al. 1973; Nault and Bowers 1974; Pickett and Griffiths 1980). Studies have shown that (E)-B-farnesene interrupts aphids from feeding and causes them to begin locomotion and often drop off the leaf in attempts to avoid predators that otherwise would devour multiple aphids (Nault and Bowers 1974; Montgomery and Nault 1977; Wohlers 1982; Pickett et al. 1992). Aphid colonies are often comprised of genetically identical individuals since a founding mother deposits clonal daughters that mature and produce additional parthenogenetic daughters (Pickett et al. 1992; Mondor and Roitberg 2000). Therefore, genes for production and detection of an alarm pheromone, while not saving the aphid preyed upon, would increase its inclusive fitness by warning related individuals nearby to escape the predator.
Aphids would maximize their fitness if all individuals in the colony could produce a uniform and optimal quantity of alarm pheromone to warn other clonal members of a predator in the vicinity. It is not known if first instar or juvenile aphids produce alarm pheromone. An increasing quantity of alarm pheromone per individual in relation to an increase in size and instar would suggest a cost of pheromone production that smaller nymphs could not afford compared to larger ones. Although producing minute quantities of pheromone requires some energy, the question in chemical ecology has been whether these costs are significant or in fact constrain amounts released. For example, bark beetles and female moths typically release ng quantities of aggregation and sex-pheromones that represent only 0.0001 percent or less of their weight (Baker 1989; Byers 1989). No studies have reported a cost of pheromone production in insects because the random variation in adult size and pheromone quantities appear to vary independently (Birgersson et al. 1988; Sappington and Taylor 1990). Only a few studies have quantified the amounts of (E)-B-farnesene in groups of aphids (Pickett and Griffiths 1980; Hedin et al. 1991) or in a few individuals (Gut and Van Oosten 1985), and no studies have reported amounts in young nymphs. The objective of this study was (1) to quantify the minute amounts of (E)-B-farnesene in individual cotton aphids, both winged and apterous (wingless), and (2) to determine the relation between amounts of pheromone and weights of individuals of a range of nymphal instars and adults from a colony on cotton plants in a glasshouse.
Materials and Methods
Cotton aphids, A. gossypii, were collected for chemical analysis from two colonies on young cotton plants (Gossypium hirsutum L.), one colony maintained in an environmental chamber at 26° C (18:6 L:D) and another in a glasshouse (28±6° C) under natural sunlight and photoperiod (11:13 L:D; Oct-Nov 2003). The aphid colonies were originally collected from the same wild population on cotton grown near Maricopa, Arizona (45 km south of Phoenix). Each colony had six young cotton plants, of which two plants in each colony were replaced weekly by plants grown previously to a height of 30-40 cm in another glasshouse.
Volatile chemicals from aphids were collected from the headspace by SPME (solid phase microextraction) and from insects by solvent extraction. For SPME, groups of about 30 adult cotton aphids were placed in 2 ml glass vials with Teflon liners and exposed to a 65 µm Carbowax-divinylbenzene fiber (Supelco, Bellefonte, PA) for 1 min before analysis by GC-MS. The GC Varian 3900 used a fused-silica capillary column (30 m x 0.25 mm ID) coated with 0.25 mm of CP-Sil 8 CB (Varian #CP5860) and helium carrier gas at a constant flow of 1.2 ml/min through the column. For SPME analyses, the injection was splitless for 5 min (thereafter split 20:1) and the oven temperature 40° C for 5 min, and then increasing by 5o per min to 180°, thereafter rising by 20° per min to 250° C and held for 5 min. Mass spectral analyses of the GC effluents were done by a Varian Saturn 2100D using the NIST02 spectral database and by comparison to a standard of (E)-B-farnesene (99%, Bedoukian).
Individuals of first instar nymphs, adults, and groups of 30 adult aphids from the environmental chamber were extracted for (E)-B-farnesene with 25, 50, or 100 µl pentane with an internal standard of ethyl heptanoate (1 ng/µl, Aldrich). Aphids were dropped into solvent contained in a 100 µl conical vial and crushed with a blunt nickel-plated tapestry needle (Prym-Dritz Corp., Spartanburg, SC) and analyzed soon thereafter by GC-MS. The extraction was efficient since a second extraction with 50 µl performed on a few individuals did not reveal additional amounts of pheromone. Individuals of various sizes also were selected from the glasshouse colony by letting them walk upon a fine paintbrush that allowed their transport to an ultra-sensitive Mettler microbalance with 0.1 µg precision for weighing prior to extraction in 50 µl pentane. The column and temperature program used for solvent extracts was the same as for SPME above, except the injection had an 0.8 min pressure pulse (45 psi) and was splitless for 0.75 min before split 60:1 for 4.25 min (thereafter 20:1), and maintained an additional 10 min at 250° C.
For quantification of (E)-B-farnesene in solvent extracts, the total GC-MS ion chromatogram (TIC) was filtered by Varian SaturnView Workstation software (version 5.52) for ions 69 and 93 to obtain peak integration areas at the retention time of the compound. The filtering of ions was deemed necessary to increase accuracy by removing TIC peaks of co-eluting contaminants (see inset Fig. 1). Areas of filtered ions and of an internal standard of ethyl heptanoate (1 ng/µl, Aldrich) were used for quantification after adjusting for the MS response factor sensitivities of the compounds. Differences between means of chemical amounts were analyzed by t-tests, and linear and non-linear regression was performed on scatter plots of aphid weights and (E)-B-farnesene amounts (Sokal and Rohlf 1995).
Results
The SPME fiber collected volatile (E)-B-farnesene from the 2 ml headspace air of groups of 30 aphids in sufficient quantities that could be identified by matching reference spectra and retention time. However, amounts could not be determined due to the semi-quantitative nature of SPME. The weight of first instar aphids reared in the environmental chamber averaged 9.5 ± 1.6 µg (±95% CL, n = 10), while larger apterous adults averaged 55.1 ± 8.3 µg (n = 9). The first instar aphids (Fig. 1) contained 0.08 ± 0.03 ng of (E)-B-farnesene, which was significantly less than larger apterous adults that had 0.42 ± 0.26 ng (t = 2.89, df 11,10, p = 0.02). There was no significant difference in the quantities of alarm pheromone between the apterous adults and winged adults (0.53 ± 0.34 ng, n = 6) from the environmental chamber colony (t = 0.59, df = 15, p = 0.56). These values yielded a ratio of average alarm pheromone quantity to insect weight of about 10 ng/mg.
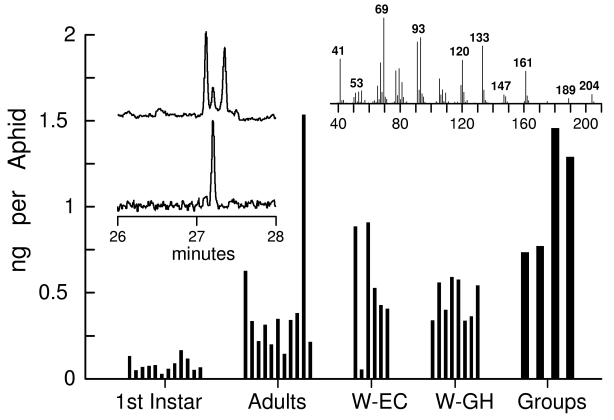
Fig. 1. Amounts of (E)-B-farnesene in individual cotton aphids, Aphis gossypii, taken from cotton plants in an environmental chamber as either first instar, apterous adults, or winged adults (W-EC) or winged adults from the glasshouse (W-GH). The inset chromatograms on left show how (E)-B-farnesene was revealed by GC-MS using the TIC (total ion current) of an aphid extract (top trace) by a filtered 69 + 93 ion sum (bottom trace) that is characteristic of the sesquiterpene. The inset at right of a mass spectrum of a GC peak at 27.2 min from an extract of 30 aphids matched (E)-B-farnesene at 0.22 ng.
To obtain a more precise relationship between aphid weight and pheromone amounts, weights of aphids from the glasshouse colony were taken prior to extraction and GC-MS. Plotting these values showed that as weight (µg) increased, the quantity of (E)-B-farnesene (ng) increased significantly and best fit a power function (Y = 0.0181X0.728, n = 67, R2 = 0.82, p < 0.001, Fig. 2). A linear function was also calculated (Y = 0.166 + 0.0033X, R2 = 0.62) for comparison to earlier studies on insects, as discussed subsequently. Winged adults from the glasshouse (0.46 ± 0.09 ng, n = 8) or environmental chamber colonies (0.53 ± 0.34 ng, Fig. 1) were not significantly different in amounts of alarm pheromone (t = 0.56, df = 12, p = 0.59). Nor were winged aphids different from apterous aphids of 50-150 µg size (0.63 ± 0.13 ng, n = 19) (t = 1.49, df = 31, p = 0.15). Although the total amounts of (E)-B-farnesene increased with weight (Fig. 2), the concentrations of (E)-B-farnesene (ng/mg fresh weight) in individual aphids declined as the body weight increased (Fig. 3). This relationship was best fit with an exponential function Y = 9.079 e- 4.365X (R2 = 0.43, p < 0.001, Fig. 3). No other sesquiterpenes or isomers of farnesene were found in the aphid extracts (detection limit of about 3 pg/aphid in group of 30).
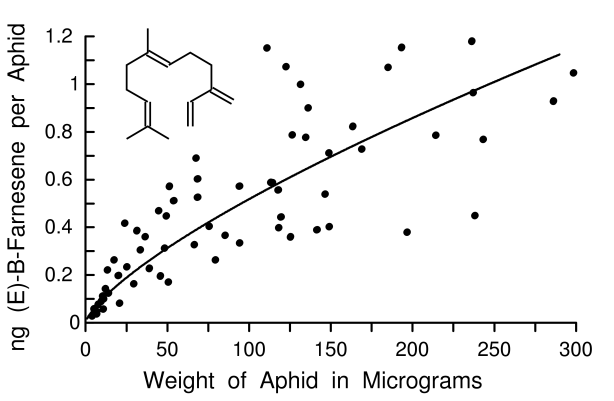
Fig. 2. Relationship between fresh weight of cotton aphid in mg and amounts of (E)-B-farnesene in ng for aphids reared in the glasshouse.
Discussion
The cotton aphid is a relatively small aphid and appears to have smaller amounts of alarm pheromone than other aphid species (Picket and Griffiths 1980; Gut and Van Oosten 1985; Hedin et al. 1991). Amounts in cotton aphids ranged up to 1.5 ng with an average of about 0.7 ± 0.1 ng in all sampled individuals weighing >50 µg (139.3 ± 20.8 µg), and somewhat higher in the four groups of large adults that averaged 1.1 ng per aphid (Fig. 1). This is the first study to show a relation between weight and alarm pheromone amounts in aphids (Fig. 2) or other insects. The six smallest nymphs (all first instar) on average weighed 6 µg, which is only 2.3 % that of the six largest adult individuals, yet the smaller ones produced proportionately more alarm pheromone in relation to their weight than did the larger adults (Fig. 3). This suggests that nymphs are trying to compensate for their smaller size in order to approach an optimal quantity for warning as would be assumed in adults. In fact, all aphids from first instar nymphs to adults had some alarm pheromone, which is consistent with every individual having an equal interest in being able to warn relatives in the colony. At a range of natural release rates, it is well established that an increase in aggregation pheromone release causes an increase in attraction distance (Byers et al. 1989; Schlyter 1992). Therefore it is reasonable to assume that with more pheromone in the body more can be released that would have a longer signaling range of beneficial warning.
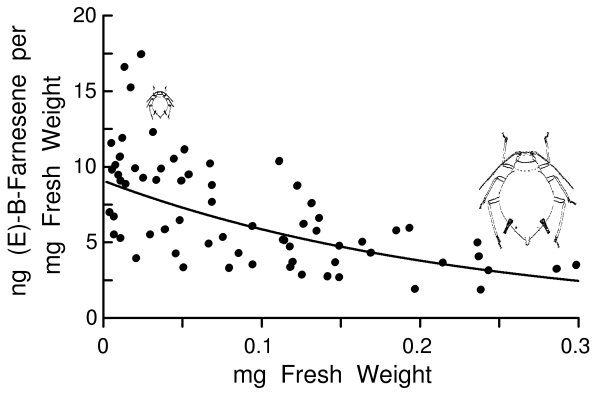
Fig. 3. Relationship between fresh weight of cotton aphid in mg and concentration of (E)-B-farnesene (ng per mg) for aphids reared in the glasshouse.
An important question in chemical ecology concerns what factors during evolution determine the level of pheromone content and emission in a species. For example, are there costs involved that allow larger individuals to afford to produce more pheromone? However, no studies have reported a significant increase in pheromone content as a function of body weight or size. Birgersson et al. (1988) took male bark beetles, Ips typographus L., attacking Norway spruce trees and found that an increase in the dry weight of males was weakly correlated with an increase in one component of its aggregation pheromone, 2-methyl-3-buten-2-ol (MB). However, variation in male weight explained only 3.2% of the variation in MB, while a second pheromone component, cis-verbenol was not correlated with size. In the butterfly Colias eurytheme, the variation in size of male forewings was not significantly correlated with amounts of three saturated hydrocarbons (heptacosane, 13-methylheptacosane, and nonacosane) of the male courtship pheromone in 6 of 7 samples, and in all samples, the variation in forewing size could explain only about 2.4% of the variation in pheromone content (Sappington and Taylor 1990). The lack of significant relationships between size and pheromone content may be explained by the fact that most pheromones are produced by adults. For example, (a) adult insects vary much less in size compared to juveniles+adults, (b) the amounts of pheromone in adults may be nearly constant due to stabilizing selection (gene fixation), or (c) there is substantial random variation in pheromone quantities due to environmental factors.
In contrast, the cotton aphids over a large range of weights of up to 60-fold produced pheromone, thus resulting in significant relationships where about 82% of the variation in alarm pheromone can be explained by variation in weight (or 62% using methods comparable to the previous studies). The increase in amounts of alarm pheromone in relation to larger size indicates there is a significant energetic cost in producing pheromone. If the costs were insignificant then all individuals, regardless of size, ought to produce similar amounts since in theory all are identically related and would benefit by evolving a specific and optimal quantity for warning. The observed relationship was probably not due to a size constraint since the smallest aphids appear able to easily hold 1 ng of pheromone, as found in larger aphids, and this volume would displace only 1/6000th of the smallest aphid's body. Therefore, alarm pheromone production in smaller aphids has a cost that probably is balanced against other costs necessary for growth and maturation, while nearly grown aphids can afford additional costs needed to produce more pheromone. Costs of pheromone production may be more easily observed in adult insects under stressful conditions. However, only insects that produce a pheromone over all life stages would be expected to show significant relationships between size and pheromone amounts, indicating biosynthetic costs that smaller individuals are unable to expend.
References
Baker, TC (1989) Sex pheromone communication in the Lepidoptera: new research progress. Experientia, 45:248-262.
Birgersson, G, Schlyter, F, Bergström, G, Löfqvist (1988) Individual variation in aggregation pheromone content of the bark beetle, Ips typographus. J. Chem. Ecol. 14:1737-1761.
Bowers, WS, Nault, LR, Webb, RE, Dutky, SR (1972) Aphid alarm pheromone: Isolation, identification, synthesis. Science 177:1121-1122.
Byers, JA (1989) Chemical ecology of bark beetles. Experientia 45:271-283.
Byers, JA, Anderbrant, O, Löfqvist, J (1989) Effective attraction radius: a method for comparing species attractants and determining densities of flying insects. J. Chem. Ecol., 15:749-765.
Edwards, LJ, Siddal, JB, Dunham, LL, Uden, P, Kislow, CJ (1973) Trans-b-farnesene, alarm pheromone of the green peach aphid, Myzus persicae (Sulzer). Nature 241:126-127.
El-Sayed, AM (2004) The Pherobase: Database of Insect Pheromones and Semiochemicals. http://www.pherobase.com.
Gut, J, Van Oosten, AM (1985) Functional significance of the alarm pheromone composition in various morphs of the green peach aphid, Myzus persica. Entomol. Exp. Appl. 37:199-204.
Hedin, PA, Phillips, VA, Dysart, RJ (1991) Volatile constituents from honeysuckle aphids, Hyadaphis tataricae, and the honeysuckle, Lonicera spp.: Search for assembling pheromones. J. Agric. Food Chem. 39:1304-1306.
Kislow, CJ, Edwards, LJ (1972) Repellent odours in aphids. Nature 235:108-109.
Mondor, EB, Roitberg, BD (2000) Has the attraction of predatory coccinellids to cornicle droplets constrained aphid alarm signaling behavior? J. Insect Behav. 13:321-329.
Montgomery, ME, Nault, LR (1977) Comparative response of aphids to the alarm pheromone, (E)-B-farnesene. Entomol. Exp. Appl. 22:236-242.
Nault, LR, Bowers, WS (1974) Multiple alarm pheromones in aphids. Entomol. Exp. Appl. 17:455-457.
Nault, LR, Edwards, LJ, Styer, WE (1973) Aphid alarm pheromones: Secretion and reception. Environ. Entomol. 2:101-105.
Pickett, JA, Griffiths, DC (1980) Composition of aphid alarm pheromones. J. Chem. Ecol. 6:349-360.
Pickett, JA, Wadhams, LJ, Woodcock, CM, Hardie, J (1992) The chemical ecology of aphids. Ann. Rev. Entomol. 37:67-90.
Sappington, TW, Taylor, OR (1990) Developmental and environmental sources of pheromone variation in Colias eurytheme butterflies. J. Chem. Ecol. 16:2771-2786.
Schlyter, F (1992) Sampling range, attraction range, and effective attraction radius: Estimates of trap efficiency and communication distance in coleopteran pheromone and host attractant systems. J. Appl. Entomol. 114:439-454.
Sokal, RR, Rohlf, FJ (1995). Biometry. 3d edition, W.H. Freeman and Company, New York.
Wientjens, WHJM, Larwijk, AC, Van Der Marel, T (1973) Alarm pheromone of grain aphids. Experientia 29:658-660.
Wohlers, P (1982) Effect of alarm pheromone (E)- b-farnesene on aphid behaviour during flight and after landing on plants. Z. ang. Entomol. 93:102-108.
© 2004 by John A. Byers
 Abstract.
The sesquiterpene, (E)-B-farnesene, is used by many aphid species as an alarm pheromone to warn related individuals of predation. Disturbed cotton aphids, Aphis gossypii Glover, released (E)-B-farnesene into the air as detected by SPME (solid phase microextraction) and gas chromatography mass spectrometry (GC-MS). Solvent extracts of cotton aphids of various life stages and weights also were analyzed by GC-MS for sums of ions 69 and 93, which discriminated (E)-B-farnesene from coeluting compounds. Aphids of all life stages and sizes reared on cotton plants in both an environmental chamber and glasshouse contained (E)-B-farnesene in amounts ranging from 0.1 to 1.5 ng per individual. The quantities of (E)-B-farnesene in aphids increased in relation to increasing body weight, and variation in individual weights explained about 82% of the variation in alarm pheromone. However, the concentrations (ng/mg fresh weight) declined exponentially with increasing body weight. These findings indicate that aphid nymphs try to compensate by producing relatively more pheromone per weight than adults but still cannot approach an evolutionary optimal load, as assumed in adults with the greatest total amounts. This suggests that young aphids need to balance costs of growth and maturation with costs of producing the alarm pheromone.
Abstract.
The sesquiterpene, (E)-B-farnesene, is used by many aphid species as an alarm pheromone to warn related individuals of predation. Disturbed cotton aphids, Aphis gossypii Glover, released (E)-B-farnesene into the air as detected by SPME (solid phase microextraction) and gas chromatography mass spectrometry (GC-MS). Solvent extracts of cotton aphids of various life stages and weights also were analyzed by GC-MS for sums of ions 69 and 93, which discriminated (E)-B-farnesene from coeluting compounds. Aphids of all life stages and sizes reared on cotton plants in both an environmental chamber and glasshouse contained (E)-B-farnesene in amounts ranging from 0.1 to 1.5 ng per individual. The quantities of (E)-B-farnesene in aphids increased in relation to increasing body weight, and variation in individual weights explained about 82% of the variation in alarm pheromone. However, the concentrations (ng/mg fresh weight) declined exponentially with increasing body weight. These findings indicate that aphid nymphs try to compensate by producing relatively more pheromone per weight than adults but still cannot approach an evolutionary optimal load, as assumed in adults with the greatest total amounts. This suggests that young aphids need to balance costs of growth and maturation with costs of producing the alarm pheromone.

